AKA Inventing a New Flavor of Energy Storage
When we start the energy unit, we usually pick up 4 flavors (kinetic, gravitational, spring, and thermal) and note that we will recognize and name other ways of storing energy when we see them. That happens pretty immediately when we do pie charts.
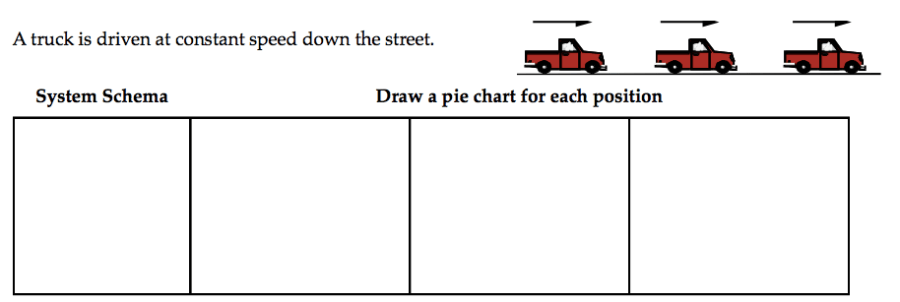
We run into a problem pretty quickly, here. The first way they try to resolve it is to make each pie entirely kinetic energy. Some students aren’t satisfied with this, but they might get convinced by their groups that there is no ∆Etherm because they just can’t see another way to make it work. Asking them how you can tell if a car was driven recently usually works. (“If you feel the hood, it will be wa… oh.”) So thermal energy must be increasing and kinetic energy must be staying constant. Something else must be getting smaller—can you drive a car at a constant speed forever? (“Sure, as long as the road keeps going and you don’t run out of… oh.”)
The gas must be storing energy somehow. And it’s not because of motion, a spring, a gravitational interaction, or warmth. So this must be a “new” way to store energy. In fuel. A chemical energy. I used to use Echem, but my students pointed out that chemical energy is an interaction energy, so it must be Uchem. Good point.
What does any of this have to do with tic tacs?
Right. So. At some point in the energy transfer unit, I will bring in some tic tacs and take a few minutes to do a quick qualitative problem with them. (Well, this is what I do now, but I’ve also used this as a quiz question in the past.) A funny thing happens when you drop a tic tac on a table. Sometimes it bounces higher on the second bounce than it does on a first bounce.

At this point, you’ll want to grab a tic tac and start dropping it. After you do it a few times, you’ll start to notice. It’s strange to see those “aberrant” bounces.
Last year, my students wanted to take a video to get a better view of what was happening. It took a few tries, but we got a pretty nice one. I suggest changing the quality to HD and watching full screen so that you can really see the difference in spinning.
These students have not studied rotational motion (and won’t study it in this class). We haven’t talked about rotational kinetic energy, but they recognize that there must be energy stored in spinning, so there must be a spinning flavor of energy. It’s a motion. It doesn’t depend on an interaction. So it makes sense for this to be a kinetic energy, not an interaction energy.
Here’s what that class ended up drawing to represent the situation. (I was trying out the TIPERs-style of energy bar charts instead of LOL diagrams. I’m planning to go back full time to LOLs in the future, but that’s a whole other story. In short: defining the system in the diagram itself is really worth it (for me).)
Another “new” flavor of energy to add to our list. It’s a pretty simple, quick, engaging, and physical-but-qualitative problem. And after seeing this problem, they start noticing other times when energy might be stored as rotational kinetic energy, adding a little more depth to the discussions of future problems.
—
Shout-outs to:
Paulette Struckman who shared this phenomenon with me (and two of her students who used it for a science fair project) in my first year of teaching.
Andy Rundquist who has a blog post focusing on a totally different aspect of this kind of bouncing (with rugby balls, not tic tacs).

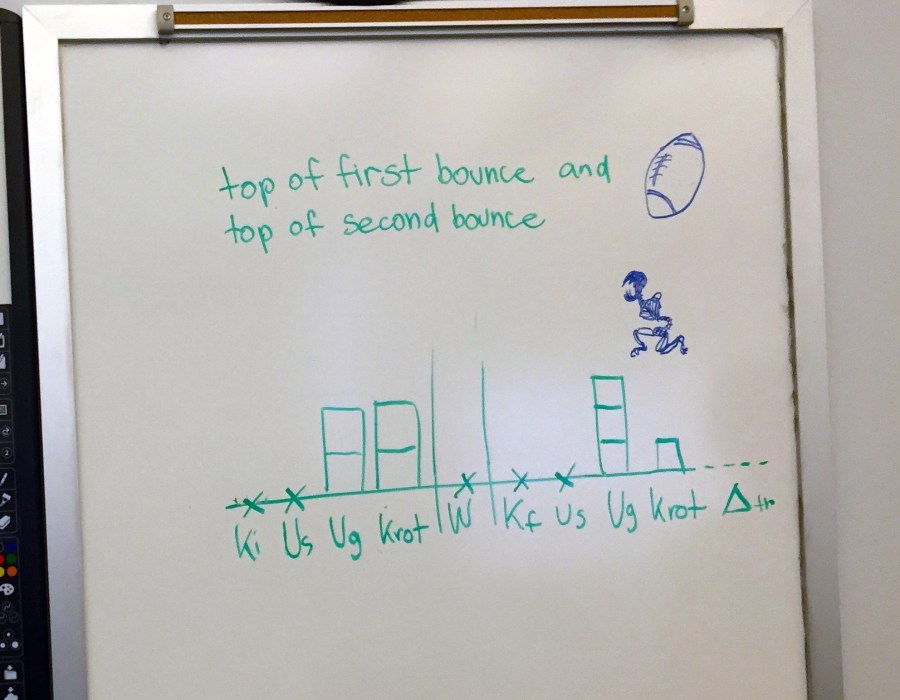

I love the fact that in that video the first bounce causes a rotation that’s hard to see (it seems that the axis of rotation is in the plane of the video).
Here’s my question: What would be wrong about lumping that rotational energy into thermal? One problem I see is that rarely do you get such organized motion (the high bounce) out of thermal energy, but this is a pretty singular system (really one particle as opposed to the N particles necessary to model thermal stuff).
I’m not 100% sure I understand what you are asking. Are you saying that we would model the energy as being stored as thermal initially and transferred to being gravitational energy instead of thinking of it as rotational kinetic energy? So basically we think of it as being colder when it is higher and warmer when it is lower?
If so, then—I would have to think more about that for myself. For my students, I think that it wouldn’t jive totally with the way we’ve been thinking/talking about thermal energy (as submicroscopic kinetic energies, or the random-direction shaking of tiny particles). I think it would be weird to say that motion we could see at the macroscopic level would be considered thermal energy. (Also, in our class thermal energy only increases. In chemistry (or when they come back for a second year of physics) things can cool off.)
Or maybe say more about what you’re thinking. I’m curious! 🙂
I guess what I was thinking is that thermal energy is often just internal rotational energy that doesn’t count toward macroscopic translation. I guess there’s a problem with thinking about macroscopic rotation, but again this is a weird singular system.
Now I’m imagining dropping a large box with tic tacs inside that I can’t see. (Not the box they came in. A bigger box.) When the box hits the ground, some of the initial energy of the system goes to making things warmer and some goes to making tic tacs spin more. Cool! If I ever get another chance at a 2nd year HS class, I’d want to explore this line of thinking more with them. Thanks!
Love it!
Andy, one concern I have with lumping rotational with thermal is I tend to use thermal as the “dumping ground” for energy that cannot be easily recovered in “usable” (e.g. kinetic or gravitational) forms. But we see that rotational clearly can be ‘recovered.’
[…] with numerical problem solving. So we started today by gathering around one of the lab tables and looking at a tic tac as it bounced. Once they started noticing what I was seeing, I gave them a bunch to use on their own, and there […]
THANK YOU FOR THIS! I just used it for my AP Physics 1 class as a demo for introducing rotational energy. After they explained why a bouncing ball bounces lower and lower on each consecutive bounce, they were a bit flummoxed to see a tic tac’s variable bounce. Sure enough after about three minutes, some students were able to notice the difference in rotation and from there we moved right into the disk vs ring race down a ramp. Thank you so much for sharing this idea and please keep the good stuff coming!
Awesome! I’m so glad it worked well for you. 🙂
[…] I’d like to make ten-sided blocks – kinetic, gravitational, elastic, electric, chemical, nuclear, thermal, electromagnetic, acoustic, and – most importantly – a MYSTERY FLAVOR side! The “none of the above” option is an incredibly important one, as you can always find new flavors of energy. […]